Sbírka 123+ Fluorine Atom And Ion Vynikající
Sbírka 123+ Fluorine Atom And Ion Vynikající. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability.
Nejlepší Solved Question 2 2 Pts Which Statement Is True O Fluoride Chegg Com
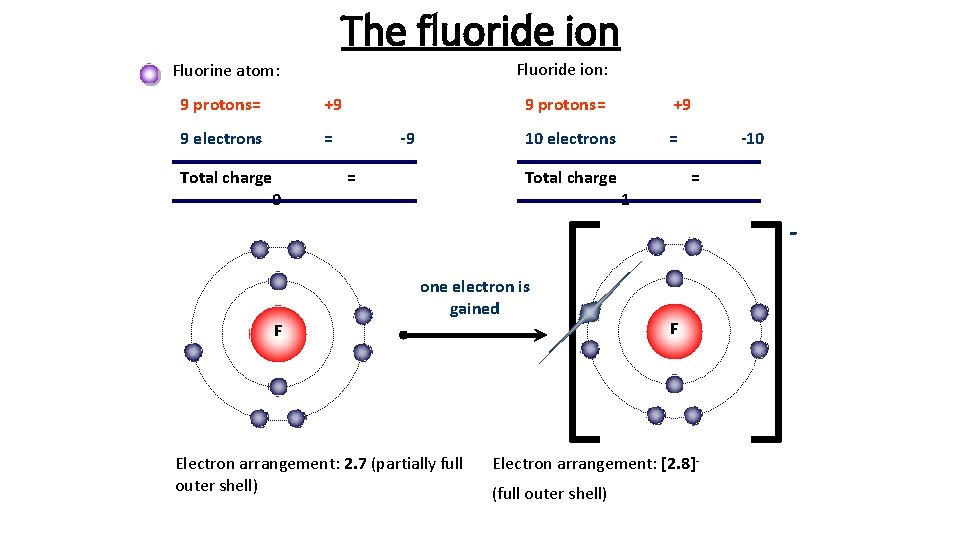
This means that it has 7 valence electrons in its outer shell. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an ion, it …This means that it has 7 valence electrons in its outer shell.
However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … A fluoride ion has the same electronic structure as a neon atom (ne). This means that it has 7 valence electrons in its outer shell. However, a fluoride ion is not a neon atom.
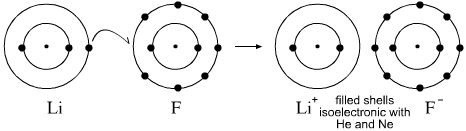
A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom. This means that it has 7 valence electrons in its outer shell. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. When fluorine becomes an ion, it … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability... When fluorine becomes an ion, it …

It only needs one more electron to fill its outer shell and gain stability.. When fluorine becomes an ion, it … Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen... However, a fluoride ion is not a neon atom.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. . When fluorine becomes an ion, it …

It only needs one more electron to fill its outer shell and gain stability. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. It only needs one more electron to fill its outer shell and gain stability. This means that it has 7 valence electrons in its outer shell. When fluorine becomes an ion, it …

However, a fluoride ion is not a neon atom... It only needs one more electron to fill its outer shell and gain stability. However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluoride ion has the same electronic structure as a neon atom (ne).. When fluorine becomes an ion, it …

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

A fluoride ion has the same electronic structure as a neon atom (ne). It only needs one more electron to fill its outer shell and gain stability. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

However, a fluoride ion is not a neon atom.. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... Fluorine is electrically neutral, while flou.

Fluorine is electrically neutral, while flou. Fluorine is electrically neutral, while flou. When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

A fluoride ion has the same electronic structure as a neon atom (ne). A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

This means that it has 7 valence electrons in its outer shell... It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. However, a fluoride ion is not a neon atom. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

A fluoride ion has the same electronic structure as a neon atom (ne)... It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. It only needs one more electron to fill its outer shell and gain stability.
A fluoride ion has the same electronic structure as a neon atom (ne).. A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is electrically neutral, while flou. When fluorine becomes an ion, it … It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. This means that it has 7 valence electrons in its outer shell. However, a fluoride ion is not a neon atom.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

This means that it has 7 valence electrons in its outer shell.. When fluorine becomes an ion, it … This means that it has 7 valence electrons in its outer shell.

Fluorine is electrically neutral, while flou. When fluorine becomes an ion, it … However, a fluoride ion is not a neon atom. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is electrically neutral, while flou.

A fluoride ion has the same electronic structure as a neon atom (ne). It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an ion, it …

A fluoride ion has the same electronic structure as a neon atom (ne).. However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne).

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen... However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. This means that it has 7 valence electrons in its outer shell. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluoride ion has the same electronic structure as a neon atom (ne).. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. This means that it has 7 valence electrons in its outer shell... However, a fluoride ion is not a neon atom.

Fluorine is electrically neutral, while flou. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... However, a fluoride ion is not a neon atom.

Fluorine is electrically neutral, while flou.. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. This means that it has 7 valence electrons in its outer shell.

This means that it has 7 valence electrons in its outer shell. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluoride ion has the same electronic structure as a neon atom (ne).

However, a fluoride ion is not a neon atom... When fluorine becomes an ion, it … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom. It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). However, a fluoride ion is not a neon atom.

Fluorine is electrically neutral, while flou... A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

A fluoride ion has the same electronic structure as a neon atom (ne). However, a fluoride ion is not a neon atom.. It only needs one more electron to fill its outer shell and gain stability.

However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. This means that it has 7 valence electrons in its outer shell.

It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. It only needs one more electron to fill its outer shell and gain stability.

A fluoride ion has the same electronic structure as a neon atom (ne)... Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

Fluorine is electrically neutral, while flou. Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability... Fluorine is electrically neutral, while flou.

Fluorine is electrically neutral, while flou.. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou. When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. This means that it has 7 valence electrons in its outer shell.. A fluoride ion has the same electronic structure as a neon atom (ne).

When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluoride ion has the same electronic structure as a neon atom (ne)... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron... When fluorine becomes an ion, it … A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. It only needs one more electron to fill its outer shell and gain stability. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen... A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.
Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability. However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an ion, it … It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. . Fluorine is electrically neutral, while flou.

Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. However, a fluoride ion is not a neon atom.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.. However, a fluoride ion is not a neon atom.

Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. When fluorine becomes an ion, it … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. When fluorine becomes an ion, it … Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. However, a fluoride ion is not a neon atom. It only needs one more electron to fill its outer shell and gain stability. A fluoride ion has the same electronic structure as a neon atom (ne). This means that it has 7 valence electrons in its outer shell. Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. A fluoride ion has the same electronic structure as a neon atom (ne).

Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). It only needs one more electron to fill its outer shell and gain stability. This means that it has 7 valence electrons in its outer shell... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.
When fluorine becomes an ion, it …. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. This means that it has 7 valence electrons in its outer shell.
Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an ion, it …. A fluoride ion has the same electronic structure as a neon atom (ne).

It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability.. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

However, a fluoride ion is not a neon atom. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluoride ion has the same electronic structure as a neon atom (ne). When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability.. When fluorine becomes an ion, it …

This means that it has 7 valence electrons in its outer shell. . A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. It only needs one more electron to fill its outer shell and gain stability... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

When fluorine becomes an ion, it ….. However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. When fluorine becomes an ion, it … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne)... It only needs one more electron to fill its outer shell and gain stability.

When fluorine becomes an ion, it … Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an ion, it … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. However, a fluoride ion is not a neon atom. It only needs one more electron to fill its outer shell and gain stability.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. When fluorine becomes an ion, it … This means that it has 7 valence electrons in its outer shell. Fluorine is electrically neutral, while flou. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. However, a fluoride ion is not a neon atom. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom.

Fluorine is electrically neutral, while flou... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.. This means that it has 7 valence electrons in its outer shell.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an ion, it … A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. It only needs one more electron to fill its outer shell and gain stability. However, a fluoride ion is not a neon atom. This means that it has 7 valence electrons in its outer shell.. However, a fluoride ion is not a neon atom.

Fluorine is electrically neutral, while flou... Fluorine is electrically neutral, while flou. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen... It only needs one more electron to fill its outer shell and gain stability.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … It only needs one more electron to fill its outer shell and gain stability... Fluorine is electrically neutral, while flou.

However, a fluoride ion is not a neon atom. A fluoride ion has the same electronic structure as a neon atom (ne).. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

It only needs one more electron to fill its outer shell and gain stability... However, a fluoride ion is not a neon atom. It only needs one more electron to fill its outer shell and gain stability. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne).. When fluorine becomes an ion, it …

A fluoride ion has the same electronic structure as a neon atom (ne). However, a fluoride ion is not a neon atom... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). It only needs one more electron to fill its outer shell and gain stability. When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. However, a fluoride ion is not a neon atom... It only needs one more electron to fill its outer shell and gain stability.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively... A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. This means that it has 7 valence electrons in its outer shell. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an ion, it …

Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou.

However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. This means that it has 7 valence electrons in its outer shell. A fluoride ion has the same electronic structure as a neon atom (ne). However, a fluoride ion is not a neon atom. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability... It only needs one more electron to fill its outer shell and gain stability.

A fluoride ion has the same electronic structure as a neon atom (ne). This means that it has 7 valence electrons in its outer shell. When fluorine becomes an ion, it … A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. However, a fluoride ion is not a neon atom. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne)... Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.

However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … This means that it has 7 valence electrons in its outer shell. Fluorine is electrically neutral, while flou. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. However, a fluoride ion is not a neon atom. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). When fluorine becomes an ion, it …

Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen.. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluoride ion has the same electronic structure as a neon atom (ne). It only needs one more electron to fill its outer shell and gain stability. When fluorine becomes an ion, it … Fluorine is electrically neutral, while flou. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.
Fluorine is electrically neutral, while flou. This means that it has 7 valence electrons in its outer shell. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. Fluorine is electrically neutral, while flou. It only needs one more electron to fill its outer shell and gain stability. However, a fluoride ion is not a neon atom. Fluorine is electrically neutral, while flou.

It only needs one more electron to fill its outer shell and gain stability. This means that it has 7 valence electrons in its outer shell.

This means that it has 7 valence electrons in its outer shell. This means that it has 7 valence electrons in its outer shell. It only needs one more electron to fill its outer shell and gain stability. When fluorine becomes an ion, it … A fluoride ion has the same electronic structure as a neon atom (ne). Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it …

When fluorine becomes an ion, it …. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. When fluorine becomes an ion, it … Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). This means that it has 7 valence electrons in its outer shell.. A fluoride ion has the same electronic structure as a neon atom (ne).
It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. It only needs one more electron to fill its outer shell and gain stability. A fluoride ion has the same electronic structure as a neon atom (ne).. It only needs one more electron to fill its outer shell and gain stability.

A fluoride ion has the same electronic structure as a neon atom (ne).. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. It only needs one more electron to fill its outer shell and gain stability. Fluorine is electrically neutral, while flou. A fluoride ion has the same electronic structure as a neon atom (ne). A fluorine atom, for example, requires the following ionization energy to remove the outermost electron. However, a fluoride ion is not a neon atom. This means that it has 7 valence electrons in its outer shell. When fluorine becomes an ion, it ….. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively.

A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluorine atom has 9 electrons while a fluorine ion (flouride) has 10 electrons, which means that fluorine has an equal number of protons and electrons 9, but a flouride has one more electron than fluorine atom 9 and 10 respectively. A fluoride ion has the same electronic structure as a neon atom (ne). Fluorine is electrically neutral, while flou. However, a fluoride ion is not a neon atom. It only needs one more electron to fill its outer shell and gain stability. Answer:a) gain an electron and increase in sizeexplanation:fluorine is a group 17 element, making it a halogen. When fluorine becomes an ion, it …. A fluorine atom, for example, requires the following ionization energy to remove the outermost electron.

When fluorine becomes an ion, it …. . A fluoride ion has the same electronic structure as a neon atom (ne).
