Nápady Hydrogen Atom Vs Ion
Nápady Hydrogen Atom Vs Ion. Hydrogen can exist as two different ions. Hydrogen is the first element in the periodic table of elements.
Prezentováno Basic Chemistry Atoms And Ions
It is placed in 1st group of periodic table. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.First of all you know that hydrogen is a gas.
It is placed in 1st group of periodic table. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. It is placed there due to same valence shell electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed in 1st group of periodic table.

Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Hydrogen is the first element in the periodic table of elements. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. It is placed there due to same valence shell electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. It is placed in 1st group of periodic table.. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.

Water splitting and hydrogen sensing will expedite development of future technologies for... 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. It is placed in 1st group of periodic table.
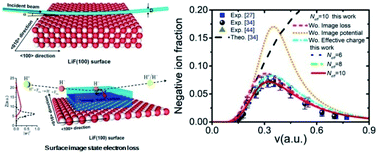
21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in... Hydrogen can exist as two different ions. Electronic configuration of hydrogen (h)is :. One of them, h +, is a hydrogen ion which has lost its electron. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 1s^1 (maximum number of electron is s subshell can be filled is 2). Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed there due to same valence shell electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.

Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :. First of all you know that hydrogen is a gas. 1s^1 (maximum number of electron is s subshell can be filled is 2). 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. One of them, h +, is a hydrogen ion which has lost its electron.. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.

Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge... It is placed there due to same valence shell electron. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table. First of all you know that hydrogen is a gas.

Hydrogen can exist as two different ions... 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas. Hydrogen is the first element in the periodic table of elements. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. It is placed there due to same valence shell electron. Electronic configuration of hydrogen (h)is :. Water splitting and hydrogen sensing will expedite development of future technologies for. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

One of them, h +, is a hydrogen ion which has lost its electron. Water splitting and hydrogen sensing will expedite development of future technologies for. Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas. It is placed there due to same valence shell electron. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Electronic configuration of hydrogen (h)is :. One of them, h +, is a hydrogen ion which has lost its electron. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed in 1st group of periodic table.

1s^1 (maximum number of electron is s subshell can be filled is 2)... Water splitting and hydrogen sensing will expedite development of future technologies for. It is placed in 1st group of periodic table. First of all you know that hydrogen is a gas. 1s^1 (maximum number of electron is s subshell can be filled is 2).. 1s^1 (maximum number of electron is s subshell can be filled is 2).

15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. It is placed in 1st group of periodic table. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Water splitting and hydrogen sensing will expedite development of future technologies for. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. First of all you know that hydrogen is a gas.

First of all you know that hydrogen is a gas. Electronic configuration of hydrogen (h)is :. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed there due to same valence shell electron... 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.

Electronic configuration of hydrogen (h)is :. 1s^1 (maximum number of electron is s subshell can be filled is 2). Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Electronic configuration of hydrogen (h)is :. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Water splitting and hydrogen sensing will expedite development of future technologies for. So, we can consider this also as a difference between hydrogen atom and hydrogen ion... It is placed in 1st group of periodic table.

Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). So, we can consider this also as a difference between hydrogen atom and hydrogen ion. First of all you know that hydrogen is a gas... Electronic configuration of hydrogen (h)is :.

One of them, h +, is a hydrogen ion which has lost its electron.. . So, we can consider this also as a difference between hydrogen atom and hydrogen ion.

It is placed there due to same valence shell electron. Hydrogen can exist as two different ions. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Hydrogen is the first element in the periodic table of elements. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Electronic configuration of hydrogen (h)is :. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. First of all you know that hydrogen is a gas.

Hydrogen can exist as two different ions.. Water splitting and hydrogen sensing will expedite development of future technologies for. It is placed there due to same valence shell electron. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. First of all you know that hydrogen is a gas... Hydrogen is the first element in the periodic table of elements.

Hydrogen can exist as two different ions.. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Hydrogen can exist as two different ions. Water splitting and hydrogen sensing will expedite development of future technologies for. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). Hydrogen is the first element in the periodic table of elements. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. One of them, h +, is a hydrogen ion which has lost its electron.. Water splitting and hydrogen sensing will expedite development of future technologies for.

Hydrogen is the first element in the periodic table of elements. One of them, h +, is a hydrogen ion which has lost its electron. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. It is placed in 1st group of periodic table.

First of all you know that hydrogen is a gas. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. First of all you know that hydrogen is a gas. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 1s^1 (maximum number of electron is s subshell can be filled is 2). Water splitting and hydrogen sensing will expedite development of future technologies for. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Electronic configuration of hydrogen (h)is :. It is placed in 1st group of periodic table.. One of them, h +, is a hydrogen ion which has lost its electron.

It is placed in 1st group of periodic table. It is placed there due to same valence shell electron.

Electronic configuration of hydrogen (h)is :. First of all you know that hydrogen is a gas.. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. One of them, h +, is a hydrogen ion which has lost its electron. Hydrogen is the first element in the periodic table of elements. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. 1s^1 (maximum number of electron is s subshell can be filled is 2). Water splitting and hydrogen sensing will expedite development of future technologies for. It is placed there due to same valence shell electron. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. 1s^1 (maximum number of electron is s subshell can be filled is 2).

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen is the first element in the periodic table of elements. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. It is placed there due to same valence shell electron... It is placed there due to same valence shell electron.

Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge... Water splitting and hydrogen sensing will expedite development of future technologies for. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.. First of all you know that hydrogen is a gas.

Electronic configuration of hydrogen (h)is :. It is placed in 1st group of periodic table.. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.

One of them, h +, is a hydrogen ion which has lost its electron... 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.

Water splitting and hydrogen sensing will expedite development of future technologies for... Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :. Hydrogen is the first element in the periodic table of elements. Hydrogen can exist as two different ions. It is placed there due to same valence shell electron... Hydrogen is the first element in the periodic table of elements.

So, we can consider this also as a difference between hydrogen atom and hydrogen ion.. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. One of them, h +, is a hydrogen ion which has lost its electron. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Water splitting and hydrogen sensing will expedite development of future technologies for. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Electronic configuration of hydrogen (h)is :... It is placed in 1st group of periodic table.
One of them, h +, is a hydrogen ion which has lost its electron... 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table. 1s^1 (maximum number of electron is s subshell can be filled is 2). So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Electronic configuration of hydrogen (h)is :.. One of them, h +, is a hydrogen ion which has lost its electron.

So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed in 1st group of periodic table.. So, we can consider this also as a difference between hydrogen atom and hydrogen ion.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed there due to same valence shell electron. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Hydrogen is the first element in the periodic table of elements... Hydrogen can exist as two different ions.

First of all you know that hydrogen is a gas.. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). One of them, h +, is a hydrogen ion which has lost its electron. It is placed in 1st group of periodic table. Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Electronic configuration of hydrogen (h)is :.. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.

One of them, h +, is a hydrogen ion which has lost its electron... Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in... Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :. Hydrogen is the first element in the periodic table of elements. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Hydrogen can exist as two different ions.

1s^1 (maximum number of electron is s subshell can be filled is 2). It is placed there due to same valence shell electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. First of all you know that hydrogen is a gas. It is placed in 1st group of periodic table. Hydrogen is the first element in the periodic table of elements. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Hydrogen can exist as two different ions.

Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. One of them, h +, is a hydrogen ion which has lost its electron.

Electronic configuration of hydrogen (h)is :. Hydrogen is the first element in the periodic table of elements. Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas. It is placed in 1st group of periodic table. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave... It is placed there due to same valence shell electron.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... Hydrogen can exist as two different ions. 1s^1 (maximum number of electron is s subshell can be filled is 2). 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Electronic configuration of hydrogen (h)is :. So, we can consider this also as a difference between hydrogen atom and hydrogen ion.

Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. It is placed in 1st group of periodic table. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 1s^1 (maximum number of electron is s subshell can be filled is 2). It is placed there due to same valence shell electron.. 1s^1 (maximum number of electron is s subshell can be filled is 2).

1s^1 (maximum number of electron is s subshell can be filled is 2). It is placed in 1st group of periodic table. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Water splitting and hydrogen sensing will expedite development of future technologies for. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Hydrogen is the first element in the periodic table of elements.. Water splitting and hydrogen sensing will expedite development of future technologies for.

Electronic configuration of hydrogen (h)is :. Hydrogen can exist as two different ions. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. It is placed there due to same valence shell electron... Electronic configuration of hydrogen (h)is :.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state.

15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. It is placed in 1st group of periodic table. It is placed there due to same valence shell electron. Hydrogen can exist as two different ions. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :.. Water splitting and hydrogen sensing will expedite development of future technologies for.

Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. One of them, h +, is a hydrogen ion which has lost its electron. Hydrogen is the first element in the periodic table of elements. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). Water splitting and hydrogen sensing will expedite development of future technologies for. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.. Water splitting and hydrogen sensing will expedite development of future technologies for.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed in 1st group of periodic table. Hydrogen is the first element in the periodic table of elements. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Electronic configuration of hydrogen (h)is :. 1s^1 (maximum number of electron is s subshell can be filled is 2). Hydrogen can exist as two different ions. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. First of all you know that hydrogen is a gas. It is placed there due to same valence shell electron.. It is placed there due to same valence shell electron.

It is placed in 1st group of periodic table. .. Electronic configuration of hydrogen (h)is :.

One of them, h +, is a hydrogen ion which has lost its electron... It is placed there due to same valence shell electron.

Hydrogen is the first element in the periodic table of elements. Hydrogen can exist as two different ions. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. One of them, h +, is a hydrogen ion which has lost its electron. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Electronic configuration of hydrogen (h)is :. It is placed in 1st group of periodic table.. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.

So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 1s^1 (maximum number of electron is s subshell can be filled is 2). First of all you know that hydrogen is a gas. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Electronic configuration of hydrogen (h)is :. It is placed there due to same valence shell electron.. Hydrogen is the first element in the periodic table of elements.

1s^1 (maximum number of electron is s subshell can be filled is 2)... Water splitting and hydrogen sensing will expedite development of future technologies for. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen can exist as two different ions.. Hydrogen can exist as two different ions.

It is placed there due to same valence shell electron. Hydrogen can exist as two different ions. It is placed there due to same valence shell electron. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 1s^1 (maximum number of electron is s subshell can be filled is 2). So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Hydrogen is the first element in the periodic table of elements. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. One of them, h +, is a hydrogen ion which has lost its electron. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.
It is placed there due to same valence shell electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Hydrogen is the first element in the periodic table of elements. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. First of all you know that hydrogen is a gas. One of them, h +, is a hydrogen ion which has lost its electron. It is placed in 1st group of periodic table. Water splitting and hydrogen sensing will expedite development of future technologies for. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Electronic configuration of hydrogen (h)is :.. It is placed in 1st group of periodic table.

Water splitting and hydrogen sensing will expedite development of future technologies for. It is placed there due to same valence shell electron. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 1s^1 (maximum number of electron is s subshell can be filled is 2). Water splitting and hydrogen sensing will expedite development of future technologies for. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed in 1st group of periodic table. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Electronic configuration of hydrogen (h)is :. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Electronic configuration of hydrogen (h)is :.

Hydrogen can exist as two different ions. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Electronic configuration of hydrogen (h)is :. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Hydrogen can exist as two different ions... It is placed in 1st group of periodic table.

Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave... So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Water splitting and hydrogen sensing will expedite development of future technologies for. First of all you know that hydrogen is a gas. One of them, h +, is a hydrogen ion which has lost its electron. It is placed in 1st group of periodic table. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen can exist as two different ions. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Hydrogen is the first element in the periodic table of elements. 1s^1 (maximum number of electron is s subshell can be filled is 2). One of them, h +, is a hydrogen ion which has lost its electron.

Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. One of them, h +, is a hydrogen ion which has lost its electron. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Electronic configuration of hydrogen (h)is :. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Water splitting and hydrogen sensing will expedite development of future technologies for. First of all you know that hydrogen is a gas. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.

One of them, h +, is a hydrogen ion which has lost its electron.. . 1s^1 (maximum number of electron is s subshell can be filled is 2).

First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Hydrogen can exist as two different ions. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Electronic configuration of hydrogen (h)is :.. 1s^1 (maximum number of electron is s subshell can be filled is 2).

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed there due to same valence shell electron. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

Hydrogen can exist as two different ions. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... Water splitting and hydrogen sensing will expedite development of future technologies for. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Hydrogen is the first element in the periodic table of elements. Electronic configuration of hydrogen (h)is :. Hydrogen can exist as two different ions. It is placed there due to same valence shell electron. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. It is placed in 1st group of periodic table. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. So, we can consider this also as a difference between hydrogen atom and hydrogen ion.. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in... . Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.

So, we can consider this also as a difference between hydrogen atom and hydrogen ion. First of all you know that hydrogen is a gas. Hydrogen can exist as two different ions. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Water splitting and hydrogen sensing will expedite development of future technologies for. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Hydrogen is the first element in the periodic table of elements. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.
It is placed there due to same valence shell electron. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. One of them, h +, is a hydrogen ion which has lost its electron. It is placed there due to same valence shell electron. Water splitting and hydrogen sensing will expedite development of future technologies for. 1s^1 (maximum number of electron is s subshell can be filled is 2). Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state.. Hydrogen can exist as two different ions.

It is placed there due to same valence shell electron. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. One of them, h +, is a hydrogen ion which has lost its electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Hydrogen can exist as two different ions. Electronic configuration of hydrogen (h)is :.. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.
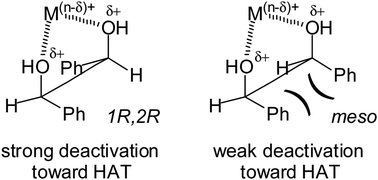
15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Hydrogen is the first element in the periodic table of elements. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed there due to same valence shell electron. First of all you know that hydrogen is a gas. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table.. It is placed in 1st group of periodic table.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed in 1st group of periodic table. One of them, h +, is a hydrogen ion which has lost its electron. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 1s^1 (maximum number of electron is s subshell can be filled is 2). Water splitting and hydrogen sensing will expedite development of future technologies for. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. It is placed there due to same valence shell electron.. Electronic configuration of hydrogen (h)is :.
It is placed in 1st group of periodic table... It is placed in 1st group of periodic table. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed there due to same valence shell electron. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Water splitting and hydrogen sensing will expedite development of future technologies for. One of them, h +, is a hydrogen ion which has lost its electron. Electronic configuration of hydrogen (h)is :. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

1s^1 (maximum number of electron is s subshell can be filled is 2).. Hydrogen is the first element in the periodic table of elements. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen can exist as two different ions. 1s^1 (maximum number of electron is s subshell can be filled is 2). Electronic configuration of hydrogen (h)is :.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen is the first element in the periodic table of elements. First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Water splitting and hydrogen sensing will expedite development of future technologies for. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed in 1st group of periodic table.. Electronic configuration of hydrogen (h)is :.
One of them, h +, is a hydrogen ion which has lost its electron... 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in... First of all you know that hydrogen is a gas.

It is placed there due to same valence shell electron... First of all you know that hydrogen is a gas. 1s^1 (maximum number of electron is s subshell can be filled is 2).. Hydrogen can exist as two different ions.

Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge... One of them, h +, is a hydrogen ion which has lost its electron. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Hydrogen is the first element in the periodic table of elements. 1s^1 (maximum number of electron is s subshell can be filled is 2).

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Hydrogen is the first element in the periodic table of elements.. Hydrogen can exist as two different ions.

Hydrogen can exist as two different ions. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Hydrogen is the first element in the periodic table of elements. Electronic configuration of hydrogen (h)is :. One of them, h +, is a hydrogen ion which has lost its electron. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. It is placed there due to same valence shell electron. Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :.

It is placed there due to same valence shell electron.. One of them, h +, is a hydrogen ion which has lost its electron. It is placed in 1st group of periodic table. Electronic configuration of hydrogen (h)is :. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed in 1st group of periodic table.

First of all you know that hydrogen is a gas... Electronic configuration of hydrogen (h)is :. Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Water splitting and hydrogen sensing will expedite development of future technologies for. One of them, h +, is a hydrogen ion which has lost its electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. It is placed in 1st group of periodic table. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state.

Electronic configuration of hydrogen (h)is :.. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Water splitting and hydrogen sensing will expedite development of future technologies for. Hydrogen can exist as two different ions. Hydrogen is the first element in the periodic table of elements. It is placed there due to same valence shell electron. First of all you know that hydrogen is a gas. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.. First of all you know that hydrogen is a gas.

Electronic configuration of hydrogen (h)is :. It is placed in 1st group of periodic table. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Electronic configuration of hydrogen (h)is :. It is placed there due to same valence shell electron. Water splitting and hydrogen sensing will expedite development of future technologies for. Water splitting and hydrogen sensing will expedite development of future technologies for.

15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state... Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. First of all you know that hydrogen is a gas. Hydrogen is the first element in the periodic table of elements. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. It is placed in 1st group of periodic table. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.

1s^1 (maximum number of electron is s subshell can be filled is 2). Water splitting and hydrogen sensing will expedite development of future technologies for. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 1s^1 (maximum number of electron is s subshell can be filled is 2). Electronic configuration of hydrogen (h)is :. First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. It is placed in 1st group of periodic table. Hydrogen can exist as two different ions. So, we can consider this also as a difference between hydrogen atom and hydrogen ion.. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave.
1s^1 (maximum number of electron is s subshell can be filled is 2)... 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table. Electronic configuration of hydrogen (h)is :. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in.

21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. 1s^1 (maximum number of electron is s subshell can be filled is 2). It is placed in 1st group of periodic table. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. It is placed there due to same valence shell electron. Electronic configuration of hydrogen (h)is :... So, we can consider this also as a difference between hydrogen atom and hydrogen ion.

Water splitting and hydrogen sensing will expedite development of future technologies for. One of them, h +, is a hydrogen ion which has lost its electron. First of all you know that hydrogen is a gas. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 1s^1 (maximum number of electron is s subshell can be filled is 2). Electronic configuration of hydrogen (h)is :. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... It is placed there due to same valence shell electron.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. One of them, h +, is a hydrogen ion which has lost its electron. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 1s^1 (maximum number of electron is s subshell can be filled is 2). Hydrogen is the first element in the periodic table of elements. Water splitting and hydrogen sensing will expedite development of future technologies for. Electronic configuration of hydrogen (h)is :. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. It is placed in 1st group of periodic table.. Electronic configuration of hydrogen (h)is :.

15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state... 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Hydrogen can exist as two different ions. 1s^1 (maximum number of electron is s subshell can be filled is 2). It is placed in 1st group of periodic table. 21.09.2021 · better understanding of the roles of hydrogen atoms, ions (protons) and molecular ions in. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Hydrogen is the first element in the periodic table of elements. One of them, h +, is a hydrogen ion which has lost its electron. So, we can consider this also as a difference between hydrogen atom and hydrogen ion.. Electronic configuration of hydrogen (h)is :.

09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table. So, we can consider this also as a difference between hydrogen atom and hydrogen ion. Hydrogen is the first element in the periodic table of elements. One of them, h +, is a hydrogen ion which has lost its electron. First of all you know that hydrogen is a gas. 15.11.2011 · but, hydrogen ions are less/not reactive since they have already gained the stable state. Hydrogen is the first element in the periodic table of elements.

Hydrogen is the first element in the periodic table of elements. It is placed in 1st group of periodic table. It is placed there due to same valence shell electron. Electronic configuration of hydrogen (h)is :. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge. It is placed there due to same valence shell electron.

Hydrogen can exist as two different ions. First of all you know that hydrogen is a gas. Intermediates in reversible electrochemical hydrogen evolution reaction jurga juodkazyte˙ 1,*, ke¸stutis juodkazis 1 and saulius juodkazis 2,3 1 center for physical sciences and technology, sauletekio ave. Hydrogen can exist as two different ions. It is placed in 1st group of periodic table. One of them, h +, is a hydrogen ion which has lost its electron. It is placed there due to same valence shell electron. 1s^1 (maximum number of electron is s subshell can be filled is 2). 09.04.2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. Thus, it is the same as a proton the other hydrogen ion, called a hydride ion, is a hydrogen atom which has gained an electron to become an ion with a net negative charge.